A healthcare device company approached Ardra Lab with a clear challenge:
“We need a compact, wrist-mounted medical device for our patented concept built on a Qualcomm SoC designed for reliability, performance, and real-world wearable constraints.”
Wearable medical devices demand far more than miniaturization. They require high-speed signal integrity, medical-grade reliability, low power performance, and a form factor that remains comfortable and durable for long-term use. Most importantly, the hardware must be engineered with precision to support advanced compute and connectivity—without sacrificing size or safety.
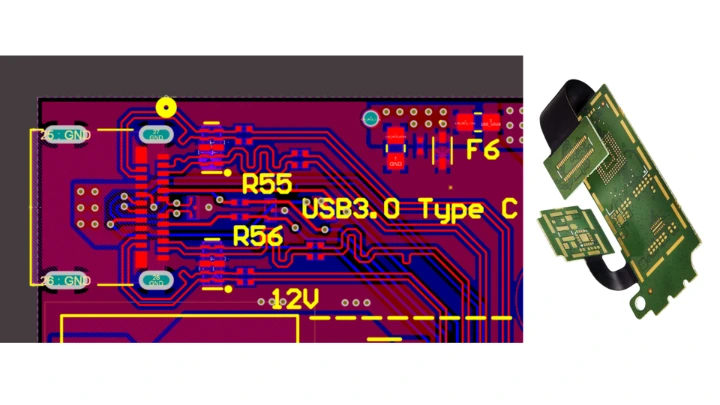
At Ardra Lab, we led the complete hardware design effort, developing a Qualcomm SoC–based PCB platform optimized specifically for wearable medical applications. We engineered high-speed, impedance-controlled, 6-layer Rigid-Flex PCBs, carefully designed to preserve signal integrity while meeting medical-grade reliability requirements.
To meet strict wearable size limits, we implemented a stacked rigid-flex architecture, enabling a highly compact, wrist-mounted form factor while maintaining strong electrical and mechanical performance. This approach ensured the product could support complex SoC-level functionality without growing bulky or compromising usability.
The result was a wearable-friendly hardware platform that combines high-speed performance, robust reliability, and compact form factor engineering—designed to meet both today’s needs and future iterations.
We are continuing development with focused efforts on further miniaturization, helping the client stay ahead of evolving medical device usability, comfort, and next-generation feature demands.
At Ardra Lab, we engineer medical and healthcare technology with compliance so your product isn’t just functional, it’s certification-ready.
Our development process is designed to align with globally recognized standards and regulatory expectations, including
ISO 13485 (Quality Management for Medical Devices),
ISO 14971 (Risk Management),
IEC 62304 (Medical Device Software Lifecycle),
IEC 60601-1 (Safety & Essential Performance), and
IEC 62366-1 (Usability Engineering). We also support regulatory deliverables and documentation frameworks required for
21 CFR, (electronic records/signatures), and
EU MDR readiness. For data-driven and connected products, we incorporate privacy and security best practices aligned with
HIPAA, GDPR, and secure-by-design principles, ensuring your solution can confidently move from prototype to clinical-grade deployment with audit-ready documentation and engineering rigor.
Have an Precision medical Product development requirement ?
Lets Connect:
Our Services Contact us